Andrea Tinelli,“Veris delli Ponti” Hospital, Scorrano, Lecce, Italy
Uterine fibroid, during its growth, cause the progressive formation of a peripheral surgical-anatomical biological structure, the myoma pseudocapsule. This anatomical entity, unknown for many years, began to have a lot of interest among gynecologists dealing with fertility surgery in the new century.
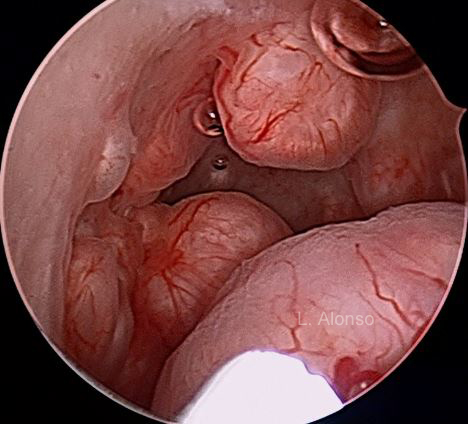
This biological structure originates from the fibroid compression of the surrounding myometrium and separates, even if in a few millimeters, fibroid from the healthy myometrium (Figure 1). In short words, pseudocapsule shifts the intact myometrial muscular fibers, like a kind of thin fibromuscular wall, maintaining the integrity and contractility of uterine musculature.
Furthermore, from the anatomical point of view, uterine fibroid is structurally anchored to its pseudocapsule by connective bridges, but it lacks its own true vascular pedicle and, occasionally, bridges of vessels and collagen fibers anchoring myoma to myometrium, interrupting the pseudocapsule surface.
From a biological point of view, the myoma pseudocapsule has the same bio-structure as the myometrium. At the ultrastructural level, visualized by transmission electron microscopy (TEM), the pseudocapsule cells have the features of smooth muscle cells like the myometrium, indicating that the pseudocapsule is part of the myometrium compressed by the myoma (Figure 2).
The most interesting biological activity that has been highlighted in the myoma pseudocapsule is that concerning the neoangiogenetic activity. In fact, myoma pseudocapsule is plentiful of collagen fibers, neurofibers and blood vessels (Figure 3), as a neurovascular bundle surrounding fibroid. Within the neurovascular bundle, some neoangiogenic factors were also studied. Angiogenetic factors identified in the pseudocapsule vessels are already widely involved in the physiology of the myometrium.
Moreover, these biochemical growth factors evaluation showed intense angiogenesis in pseudocapsule vessels and these substances are thought to have a pivotal role in wound healing and in muscular innervation. Hence, growth factors present in the myoma pseudocapsule induce angiogenesis peripherally to myometrium.
From here, therefore, we went to study a very important phase of the physiology of the uterus, wound healing, which is also based on muscular innervation. The myometrial wound healing is an interactive, dynamic process based on neuromodulators, angiogenetic factors, neuropeptides, blood cells, extracellular matrix, and parenchymal cells that follows three complex and overlapping phases: inflammation, tissue formation, and tissue remodeling. In the physiology of these processes, they also fit also nervous system and its neurotransmitters, as Substance P (SP), Vasoactive Intestinal Peptide (VIP), neuropeptide Y (NPY), Oxytocin (OXT), Vasopressin (VP), PGP 9.5, calcitonin gene-related peptide (CGRP), growth hormone-releasing hormone (GHRH). They play a role in mediating inflammation and wound healing, involved in physiology and scar repair in different tissues, including uterine muscle.
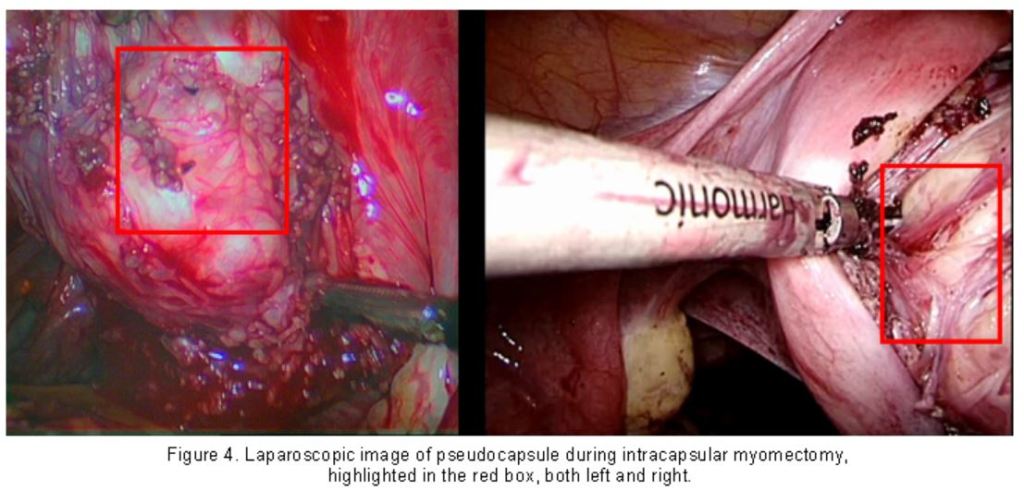
In regenerative processes associated to pseudocapsule sparing (Figure 4), as in post-myomectomy uterine scar, neuropeptides and neurotransmitters are speculatively involved in wound healing and myometrial regeneration.
The intracapsular myomectomy is based on muscular physiology and respecting anatomy, and it can be done by laparotomic, laparoscopic, robotic, vaginal and hysteroscopic approach. It can be safely done also during cesarean section, by a cesarean myomectomy.
The surgical benefit is visible during and after myomectomy: the bleeding is reduced, the myometrial anatomy is largely respected, the myometrial healing is preserved and enhanced, as confirmed by clinical and ultrasound investigations on scar site after intracapsular myomectomy.




Excellent Tinelli
LikeLike