Luis Alonso Pacheco. Centro Gutenberg. Málaga. Spain
INTRODUCTION
Adenomyosis is defined as the presence of ectopic endometrial tissue inside the myometrium. This accumulation of endometrial glands and stroma in the myometrium causes hypertrophy of the surrounding myometrium.
It was described for the first time by Rockitansky in 1860 when observing the existence of glands and endometrial cells inside the myometrium and which he called “adenoid uterium cystosarcoma” (1). It was a few years later, in 1925, when Frankl first used the term “adenomyosis” and described the presence of direct communication between the endometrial islets located in the myometrial thickness and the endometrium (2). Finally in 1972 Bird more clearly defined adenomyosis as a “benign invasion of the myometrium by the endometrium” (1)

PATHOGENESIS
Various theories have been proposed as to the origin of adenomyosis, however the true cause remains unknown. The most widely accepted theory is that adenomyosis occurs as a consequence of tissue trauma that, after a poor repair process favors invagination of the endometrium on the injured tissue (3). Another theory supports that adenomyosis is the consequence of a process of metaplasia on remaining pluripotent cells of the Müllerian ducts.
Although the true origin remains unknown, certain risk factors associated with the development of this pathology are known. Several studies have found an association between the number of pregnancies and the presence of adenomyosis, with a greater trend in those patients with a history of abortion (4).
Although adenomyosis can also appear in young patients, there appears to be an increased risk in older patients. Other known risk factors include previous uterine surgery or tamoxifen treatment.
TYPES
There is still no consensus on the depth that endometrial penetration must reach to define it as adenomyosis, although the most accepted cut-off point is 2.5mm below the myometrial endometrial junction. It is important to remember at this point that the endometrium rests directly on the myometrium at the level of the endometrial-myometrial junction or junctional zone (JZ), with an absence of mucous basement membrane.
Adenomyosis is divided into several types
Focal: Adenomyosis affects a certain area of the myometrium and can resemble a myoma, hence the term “adenomyoma”. Obviously, this adenomyoma does not have a pseudocapsule that separates it from the healthy myometrium and the vascularization is diffuse throughout the lesion and not peripheral as in the case of fibroids.
Diffuse: Affecting the entire uterine wall and causing an increase in the size and volume of it or even the entire uterus. This diffuse adenomyosis tends to affect the posterior uterine wall more frequently, then less frequently the anterior wall and rarely the coronal areas or areas close to the cervix (1)
Cystic: The term cystic adenomyosis is reserved for those cases, focal or diffuse, in which cysts with blood content greater than 1 cm in diameter are appreciated. There is a variant that is “juvenile cystic adenomyosis” that affects young women under 30 years of age in whom the adenomyosis cyst cause severe dysmenorrhea.

PREVALENCE
It is difficult to estimate the true prevalence due to the lack of well established diagnostic criteria as well as the bias that occurred years ago when studying only hysterectomy specimens and therefore in an older population.
Based on hysterectomy specimens the prevalence varies between 20-30%, although there is great variation between the different studies, with the data varying between 5-70%. Different studies based on imaging techniques established an approximate incidence of between 20-30% (5).
Of all cases of adenomyosis, approximately 30% affect women under 30 years of age, so we can say that this pathology can affect women throughout their reproductive life
CLINICAL COURSE
Patients with adenomyosis have different symptoms, including dysmenorrhea, abnormal uterine bleeding, and infertility. Not all women with adenomyosis are symptomatic and the intensity of symptoms varies according to the type and extent of adenomyosis
Abnormal uterine bleeding: It is the most frequent symptom and can affect up to 50% of patients. The presence of hypervascular areas, an increase in the endometrial surface derived from the increase in uterine size, together with dysfunctional contractility seem to be the causes of this symptom.
Dysmenorrhea: Affects approximately 30% of patients. Different theories have been proposed to explain why adenomyosis causes dysmenorrhea, it seems that the existence of uterine hypercontractility together with an increase in menstrual flow may be the triggering factors. It should be remembered that endometriosis coexists with endometrial adenomyosis in up to 24% of cases, and endometriosis may also contribute to dysmenorrhea.
Infertility: Although classically no attention has been paid because it was diagnosed mainly in multiparous women, the existence of a direct relationship between adenomyosis and infertility has been demonstrated, ruling out the existence of concomitant endometriosis (6). The cause of this association remains unknown, although it could be due to abnormal uterine peristalsis as well as associated immunological changes. We should also remember the role that associated endometriosis can play in patients with infertility.
Chronic pelvic pain and dyspareunia: These are less common symptoms.
DIAGNOSIS
The diagnosis of this pathology is based on the medical history and the gynecological exam. But today imaging studies play a capital role in diagnosis, establishing the definitive diagnosis with histopathological confirmation.
1- 2D Ultrasound: The different ultrasound patterns suggestive of adenomyosis were established in a consensus document by the MUSA group (Morphological Uterus Sonographic Assessment) and consist of the following:
A. Asymmetric thickening of the uterine walls
B. Presence of cysts in the myometrial thickness, hypoechogenic islets, shadowing, sub-endometrial echogenic lines and stippling
C. Translesional vascularization, irregular endometrial-myometrial transition and interrupted endometrial-myometrial transition. The accuracy of the diagnosis with these criteria is greater than 90%. Asymmetric thickening, sub-endometrial hyperechogenic lines, and interruption of the endometrial-myometrial transition seem to be the most accurate markers for diagnosis by 2D ultrasound (7).
2-3D Ultrasound: 3D Ultrasound allows obtaining a coronal plane in which the endometrial / myometrial interface can be observed with detail, also called junctional zone (JZ), which appears as a hypoechoic area surrounding the endometrial cavity. It has been noted that an irregular junctional zone with increased thickness constitutes a high precision ultrasound criterion for the diagnosis of adenomyosis.
3- Nuclear Magnetic Resonance Imaging (MRI): For years it has been considered the most accurate technique in the diagnosis of adenomyosis although today, many authors consider its diagnostic precision similar to that of 3D ultrasound (8). The main diagnostic criterion is the presence of endometrial foci in the thickness of the myometrium. This sign is considered pathognomonic although it only appears in 50% of cases. Other indirect signs are the presence of hyperintense linear striations that run from the basal to the myometrial thickness, thickening of the Junctional zone greater than 12mm, visualization of a globular and enlarged uterus and poor definition of the junctional zone with irregular areas.
4- Hysteroscopy: Hysteroscopy is considered the gold standard for the diagnosis of intracavitary pathology. Although adenomyosis affects the myometrium and not the endometrium, there are a series of hysteroscopic patterns highly suggestive of adenomyosis, such as the existence of irregular endometrium with surface defects, the existence of hemorrhagic cystic lesions with dark blood inside, and the existence of fibrous-looking areas (9). Hysteroscopy also allows obtaining biopsies that facilitate the establishment of a definitive diagnosis.
TREATMENT
Although traditionally the treatment of patients with adenomyosis and associated symptoms has been hysterectomy, in recent years this trend has changed and more conservative treatments are often performed. This has happened for two main reasons, on the one hand the efficacy of different medical treatments in improving the symptoms of patients with adenomyosis, and on the other hand, thanks to imaging techniques it has been observed that this pathology also affects young patients with desire to preserve future fertility in which hysterectomy cannot be considered as a viable form of treatment.
Hormonal treatments include both combined oral contraceptives such as the levonorgestrel-releasing IUD or continuous progestin treatment (OCP). Both act on the endometrium producing a decrease in menstrual bleeding and an improvement in pain. A randomized study that compared the efficacy of both, found that LNG-IUD produced a greater decrease in pain and bleeding than OCP. Continuous progesterone induces a decidualization of the endometrium along with endometrial atrophy. Improving the symptomatology of these patients.
The use of GnRH analogs is temporary option. These analogs induce suppression of ovarian function and therefore induce an hypoestrogenic state. The use of GnRH analogs is more frequent in patients who undergo assisted reproductive technique in which a decrease in the activity of adenomyosis is required since the use of analogs suppresses the inflammatory reaction in the tissues and angiogenesis, therefore decreasing the degree of adenomyosis activity.
Other non-surgical options include uterine artery embolization, which has been shown to be effective in patients with abnormal uterine bleeding and dysmenorrhea (10). Ultrasound-guided or resonance-guided ultrasound ablation with similar rates of efficacy and the use of radiofrequency ultrasound. The three techniques pursue the destruction of the adenomyoma tissue, thus improving the symptoms.
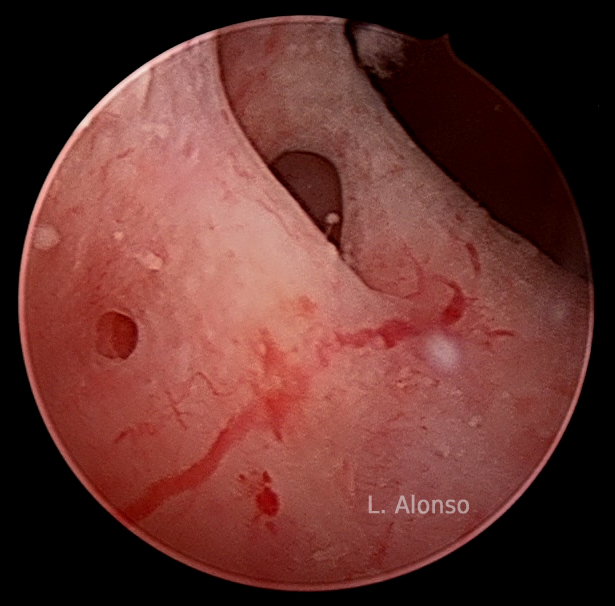
Conservative surgical techniques include effective hysteroscopic drainage of adenomyosis cysts, especially improving associated dysmenorrhea, effective endometrial ablation or resection for the treatment of abnormal uterine bleeding, and cytoreductive surgery for focal adenomyosis, which is a complex surgery with promising results only in expert hands.
CONCLUSION
Adenomyosis is an enigmatic condition both in its origin and in its treatment. The management of this pathology depends above all on the desire of future fertility of the patients. Today, the most conservative treatments are those that should be used as the first option, reserving surgical techniques as second line alternatives.
REFERENCES
1- Garcia L, Isaacson K. Adenomyosis: review of the literature. Journal of minimally invasive gynecology. 2011;18(4):428-37.
2- Bird CC, McElin TW, Manalo-Estrella P. The elusive adenomyosis of the uterus–revisited. American journal of obstetrics and gynecology. 1972;112(5):583-93.
3- Ferenczy A. Pathophysiology of adenomyosis. Human reproduction update. 1998;4(4):312-22.
4- Vercellini P, Parazzini F, Oldani S, Panazza S, Bramante T, Crosignani PG. Adenomyosis at hysterectomy: a study on frequency distribution and patient characteristics. Human reproduction. 1995;10(5):1160-2.
5- Chapron C, Tosti C, Marcellin L, Bourdon M, Lafay-Pillet MC, Millischer AE, et al. Relationship between the magnetic resonance imaging appearance of adenomyosis and endometriosis phenotypes. Human reproduction. 2017;32(7):1393-401.
6- Barrier BF, Malinowski MJ, Dick EJ, Jr., Hubbard GB, Bates GW. Adenomyosis in the baboon is associated with primary infertility. Fertility and sterility. 2004;82 Suppl 3:1091-4.
7- Accuracy of new sonographic markers in the diagnosis of adenomyosis. Dottorato di ricerca in Scienze Mediche Generali e Dei Servizi. Letizia Zannoni. 2017
8- Larsen SB, Lundorf E, Forman A, Dueholm M. Adenomyosis and junctional zone changes in patients with endometriosis. European journal of obstetrics, gynecology, and reproductive biology. 2011;157(2):206-11.
9- Di Spiezio Sardo A, Calagna G, Santangelo F, Zizolfi B, Tanos V, Perino A, et al. The Role of Hysteroscopy in the Diagnosis and Treatment of Adenomyosis. BioMed research international. 2017;2017:2518396.
10- Dueholm M. Minimally invasive treatment of adenomyosis. Best practice & research Clinical obstetrics & gynaecology. 2018;51:119-37.

0 comments on “Adenomyosis: A Review”